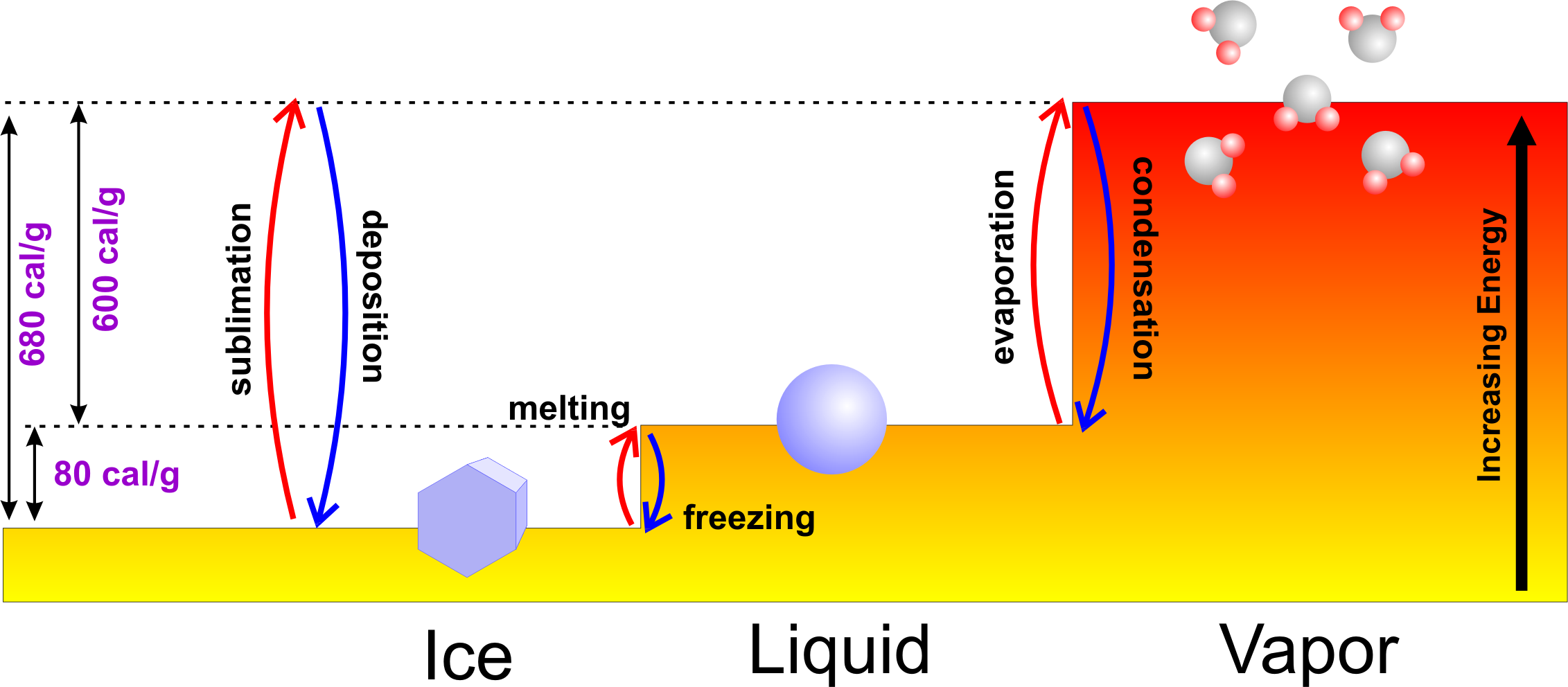
There's merit in all those ideas, but the problem is that experiments over the years have controlled for all these effects, and the results have been frustratingly inconsistent. The currents are greater in warm beakers, and could affect cooling rates. These currents occur because water cools primarily from its surface and the sides of the beaker, causing cold water to sink and warm water to rise up and take its place. Convection: Finally, there's the idea that warm water might cool faster due to increased convection currents.Evaporation: The hot water beaker loses more water molecules through evaporation, so there's less of it to freeze.So maybe warm water experiences less supercooling than cold water. This occurs because ice needs a nucleation site, such as an air bubble or impurity in the water in order to form. Supercooling: We all know that water freezes at zero degrees Celsius, but sometimes it gets a lot colder before it freezes - a phenomenon known as supercooling.

Dissolved gasses: There are more dissolved gasses in cold water than warm water, and researchers have predicted that this could play a role in cooling rates, although it's not clear how.Frost melting: Frost is an insulator, and so frosty cold water might keep its heat better than a warm beaker that melts the frost off its sides.Sir Frances Bacon and Descartes also noted the phenomenon in their studies.īut what's the physics behind this strange phenomenon?Īs the video above explains, there are five proposed mechanisms for what's going on here: It was the first peer-reviewed study on the effect, but as Derek notes in the video above, observations date all the way to Aristotle in the 4th century BCE, who observed that hot water cooled sooner than cold during his experiments.
/block-of-ice-200537317-002-57688abe5f9b58346a1b6dba.jpg)
Intrigued by this phenomenon, he went on to work with physics professor Denis Osborne, and together they were able to replicate the findings and publish a paper in 1969 showing that warm water freezes faster than cold water.
